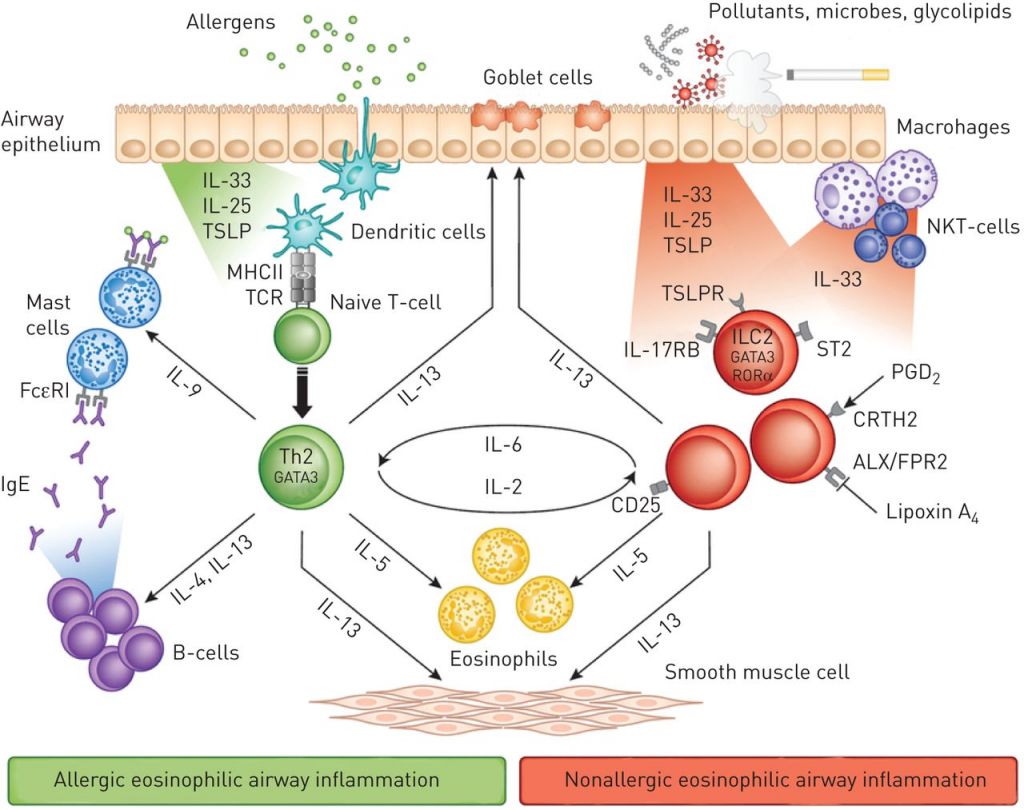
Home-based intravenous treatment with reslizumab for severe asthma in the Netherlands – An evaluation - Respiratory Medicine

Reslizumab for inadequately controlled asthma with elevated blood eosinophil counts: results from two multicentre, parallel, double-blind, randomised, placebo-controlled, phase 3 trials - The Lancet Respiratory Medicine

Cinqair (reslizumab) Injection for treatment of Severe Asthma and an Eosinophilic Phenotype - Clinical Trials Arena

Safety of Reslizumab in Uncontrolled Asthma with Eosinophilia: A Pooled Analysis from 6 Trials - The Journal of Allergy and Clinical Immunology: In Practice